As modern marine scientists, we spend a lot of time learning to effectively communicate science to various audiences. That’s mostly so we can make friends at parties, but has some nice side benefits like educating the general public, whatever. Sometimes though, and I’m probably admitting to a cardinal sin of outreach here, there’s a certain satisfaction in completely baffling your audience. When confronted with some mild nemesis, like that terrible boss from your summer job scooping ice cream, it’s kind of fun to tip your head up, assume a casual air, and trot out as many seven-syllable words as you can summon about your current research. Yeah, it would probably be better for the good of society if you didn’t just blind him with science, but let’s be honest, grad students can use an ego boost every now and then.
Pride goes before a fall though, so before you break out this strategy, pro tip: make sure you actually know what those seven-syllable words mean. Otherwise, after you’ve rattled off that you study biogeochemical cycling in Crassostrea virginica using a membrane-inlet mass spectrometer, that ice cream parlor boss might tilt his head and ask, “What’s biogeochemistry?” “Well, it’s … biology, geology, and chemistry?” you reply, feeling about as eloquent as that time he found you making out with your cute coworker in between shifts. Let’s avoid that! Welcome to your crash course on biogeochemistry.

“What IS biogeochemistry, and where are my sprinkles?!” Image from http://www.getreading.co.uk/news/local-news/ice-cream-man-its-much-4191589
The scope of “biogeochemistry” appears self-evident: it’s some frightening mishmosh of all the sciences except for physics, right? Not quite. Woods Hole Oceanographic Institution has a good definition: “Biogeochemistry is a relatively new scientific discipline that explores the physical, chemical, biological, and geological processes and reactions that govern the composition of and changes to the natural environment.” To understand why we’re throwing physics into the pot, it helps to unpack that definition a bit. Living organisms are assortments of elements, the top six of which are C, H, O, N, P, and S. Other elements, such as Ca and Mn, are also necessary for life, but are known as “trace elements” because they’re required in much smaller quantities. According to this perspective, the living world is one big beaker of elements, and the interaction of those elements can be roughly summed up as chemistry.
The business of being alive means that elements are constantly on the move, and biogeochemists are interested in tracking that movement. They’re not following particular atoms per se, but rather trying to understand the distribution of certain elements on a chosen scale. Let’s use carbon to illustrate. Carbon might be the most widely-studied element: it’s found in all living organisms and has received particular attention for its role in the atmosphere. Imagine that a scientist is researching the relationship between atmospheric carbon dioxide and marine phytoplankton in the Indian Ocean. The scientist might start by considering that phytoplankton take up CO2 from the atmosphere during photosynthesis. The phytoplankton incorporate some of that C into their biomass, but they eventually will die. (Hope you weren’t too attached.) As they sink, the dead phytoplankton may be eaten by zooplankton, who will assimilate some C into their own tissue and return some C back to the atmosphere as CO2 through respiration. Other phytoplankton will sink to the very bottom of the ocean, where they will be buried. In a few million years, those phytoplankton and the C they contain will be compressed under layers of muck and transformed into oil. That oil may be tapped by a rig and burned as fossil fuels, releasing CO2 back to the atmosphere. Phew! What seemed like a simple question of carbon in phytoplankton is revealed to be a multimillion-year odyssey, following C from critters to the seafloor to the air. That’s why every scientific discipline is needed to study biogeochemistry; if a scientist had stuck to biology, she would have ignored crucial parts of the story. And even though it goes unacknowledged, physics undergirds everything that happens as elements cycle around.
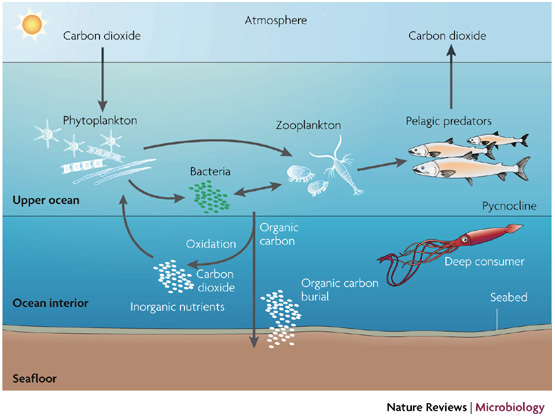
There are a few extra steps here, but you can follow phytoplankton taking up CO2 and being eaten by organisms that eventually release CO2. Some phytoplankton sink and are buried (“Organic carbon burial”). Image from a paper by Falkowski & Oliver 2007- http://www.nature.com/nrmicro/journal/v5/n10/fig_tab/nrmicro1751_F2.html
In oceanography, biogeochemistry is closely related to ecology. The similarity is hopefully evident given the above example; in both disciplines, once you start tugging at one thread, you find it’s connected to the entire quilt. Biogeochemical questions have been studied for a long time, but the formal recognition of the field is relatively recent, possibly reflecting science’s growing emphasis on interdisciplinarity. Some of the biggest questions in oceanography, such as ocean acidification, nutrient loading, and carbon sequestration, are essentially biogeochemical. This is largely because anthropogenic activity has upset the normal biogeochemical balance, creating Major Issues. The Haber-Bosch process, for example, enabled industrial N fixation, meaning that N can be added to agricultural soils in quantities not achievable by natural processes. The biogeochemical cycle of nitrogen is out whack, with too much N in a form that can be used by plants. The crops are happy, but when that extra N washes into local lakes, the end result is algae blooms and disgusted swimmers.
In fact, this explanation might make your ice cream parlor boss as happy as the crops. Not only will he have learned about biogeochemistry, but he knows that disgusted swimmers spend less time in the water and more time on land, where they will undoubtedly want ice cream. Ca-ching! Biogeochemistry really does connect everything!
For more info:
NOAA National Estuarine Research Reserve System: http://nerrs.noaa.gov/doc/siteprofile/acebasin/html/envicond/biogeo/bgtext.htm
If you’re feeling really nerdy: Biogeochemistry: An Analysis of Global Change, 2nd Edition by William H. Schlesinger. (c) 1997. San Diego, CA: Academic Press.
Cover image from http://rebloggy.com/post/uploads-sun-beach-sand-ocean-ice-cream-vertical-cone/26125737989